The New York Department of Health describes ammonia gas as a colorless, alkaline gas comprised of nitrogen and hydrogen (NH3) that has a strong odor, often associated with window cleaner. Ammonia is a natural, biological agent in organisms that helps to form amino acids, which are the basic building blocks of proteins. It also involved in the natural decomposition of plant and animal materials. When present in higher concentrations, ammonia gas is hazardous to workers and the public.
WHAT IS AMMONIA GAS USED FOR?
Many industries make use of ammonia gas for several applications:
- Cleaning chemicals
- Agricultural fertilizer
- Refrigerant gas that has in large part replaced chlorofluorocarbons (CFCs)
- Water purification
- Manufacturing plastics and other chemicals
- A building block for pharmaceuticals
AMMONIA GAS PROPERTIES
Ammonia gas is very hydrophilic, meaning it is water-loving. When stored as a gas or compressed liquid without presence of water moisture, ammonia is referred to as anhydrous ammonia.
IMPACT ON SAFETY
Upon release to the environment, ammonia gas is very quick to attach itself to moisture, such as found in a person’s eyes, mouth, throat, lungs and on his or her skin. Ammonia is very caustic, forming ammonium hydroxide that has a higher pH than water and can damage respiratory systems, disrupt vision and irritate or burn skin upon contact. The caustic action of ammonium hydroxide damages cell membranes, causing more liquid to be released that further interacts with ammonia gas, perpetuating the effects on the human body.
ODOR THRESHOLD OF AMMONIA GAS
AMMONIA GAS EXPOSURE
According to Alberta Agriculture and Forestry, nose and throat irritation can result from ammonia exposure ranging from 24–50 parts per million (ppm) after ten minutes of exposure. With a higher ammonia concentration ranging from 72–134 ppm, the same irritation can occur in half the time. For a concentration of 700 ppm, immediate and severe irritation would likely occur. At a concentration of 5,000 ppm, respiratory spasms and rapid suffocation occurs. At 10,000 ppm, pulmonary edema and potentially fatal accumulation of liquid in the lungs would occur.
DETECTION OF AMMONIA GAS
As with all safety regulations, the safe work exposure limits for ammonia vary from region to region.
- In the United Kingdom, the Health and Safety Executive specifies safe exposure limits for ammonia gas as 25 ppm for an eight-hour time-weighted average (TWA) and 35 ppm for a 15-minute short-term exposure limit (STEL).
- In the United States, OSHA specifies the permissible exposure limit (PEL) for ammonia gas as 50 ppm for an eight-hour TWA, allowing a higher limit of 100 ppm for a shorter four-hour TWA.
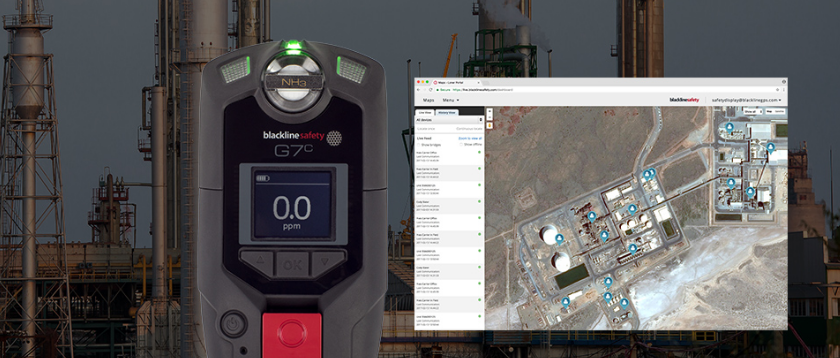
G7 WIRELESS GAS AND LONE WORKER MONITORING
While ammonia gas has a broad range of uses, from a refrigerant and fertilizer to being a raw material for many processes, it’s hazardous to the health of nearby workers and the public. Personal gas detection with an ammonia sensor is an invaluable method for businesses to help personnel keep an eye on safe work environments and the potential for ammonia gas exposure.
Let us know if you have any questions on how the G7 may be able to complement your gas detection or lone worker monitoring programs. [email protected] or (778) 686-5799





 RSS Feed
RSS Feed